AdvantaPure
BioClosure Sterile Media Bottle Assemblies
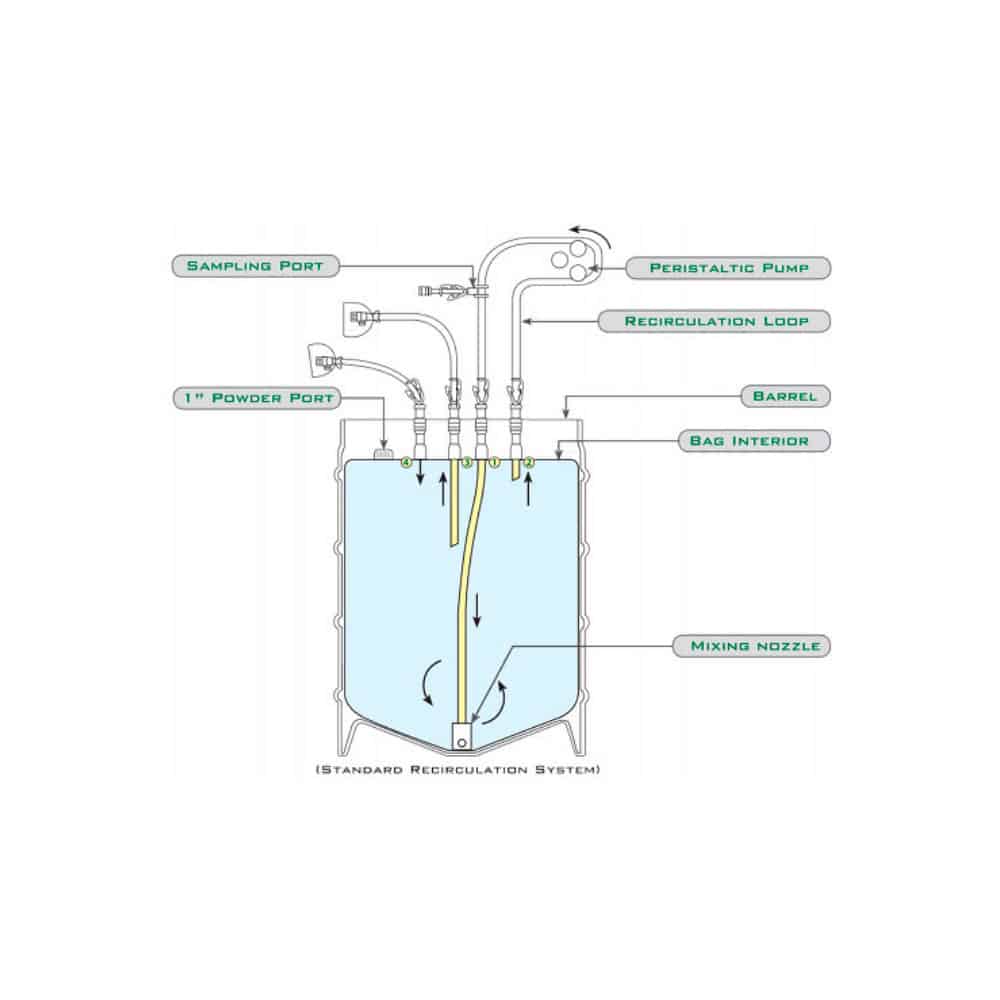
AdvantaPures sterile BioClosure media bottle assemblies
- Flexible solutions with unique customised designs and bottle choices
- Easy to customise and available in both AdvantaFlex and Silicone
- ISO 7 cleanroom produced and packaged
- Available with sterility assurance SAL 10-6
Product Number:ADVANTAPURE_BC-BOTTLE
Flexible production and open architect design make it easy to put together and meet our customers’ processes and precise design wishes.
AdvantaPure specialises in quality tubings and innovative single-use solutions for the biopharmaceutical industry. In AdvantaPure, all employees are co-owners of the company and this, combined with the close family bond, ensures our customers an extraordinary experience and service - every time.
Certificates
We offer a comprehensive documentation package on AdvantaPures BioClosure assemblies with the following certificates and documentation.
Contact us if you have questions or specific wishes and needs for documentation.
USP88
USP788
ADIF
USP Class VI
CoA
Extractable & Leachable Data
FDA
Gamma Irradiation & Sterility Assurance Level SAL 10-6
Produced in min. ISO Class 7 Cleanroom
TSE/BSE Statements
USP85
USP87
Do you have questions about our BioClosure media bottle assemblies?
We are available if you have questions or want to hear more about our BioClosure bottle assemblies, which we can adapt to your applications.

Silvia Roma Bentin
Internal Technical Sales
Popular products
Contact
Contact a specialist for more information
You are always welcome to contact us if you want to hear more about how we can design and manufacture BioClosure sterile media bottle assemblies, designed for your applications.
Our BioClosure solutions can be delivered with molded joints - hear more about all the benefits of a molded design and how it can create value for your process.