Environmental monitoring for microbial growth
Environmental monitoring is a critical component of the Good Manufacturing Practice (GMP) guidelines, especially in cleanroom settings where the control of microbial contamination is vital to ensuring the safety and quality of pharmaceutical products. Various techniques, such as air sampling, surface monitoring are employed to monitor the environment for microbial growth, each serving a specific purpose in the comprehensive strategy to maintain cleanliness and ensure the integrity of the manufacturing process.
Environmental monitoring for microbial growth is an essential part of maintaining the desired cleanliness levels in cleanroom environments, directly impacting the integrity of the manufacturing process and the quality of the final pharmaceutical products.
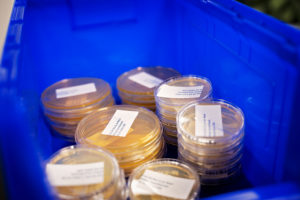
Utilizing agar plates for air, settle, and surface samples
Agar plates are a fundamental tool in the environmental monitoring of cleanrooms, enabling the detection and quantification of microbial contamination. They are used in various methods to collect samples from the air, surfaces, and even personnel, providing crucial data to ensure that the cleanroom environment meets the stringent requirements necessary for pharmaceutical, biopharmaceutical, and medical device manufacturing.
Utilizing agar plates for air, settle, and surface samples in cleanroom settings is a critical component of environmental monitoring programs. This methodology helps ensure that the cleanroom environment remains within acceptable limits of microbial contamination, safeguarding the manufacturing process and the quality of the pharmaceutical products produced.
In the meticulous environment of our cleanroom, where minimizing contamination is paramount, the utilization of agar plates plays a pivotal role in monitoring microbial growth. By incorporating agar plates into our comprehensive environmental monitoring strategy, we adeptly capture and analyze a wide range of microbial samples. This methodical approach includes making air samples, utilizing settling plates for passive air sampling, and employing contact plates for direct surface sampling.
Differential-pressure monitoring in cleanrooms
Differential-pressure monitoring is a critical aspect of maintaining the controlled environment of a cleanroom. It involves measuring and controlling the pressure difference between two areas to ensure that air flows from cleaner areas (higher pressure) to less clean areas (lower pressure). This principle is fundamental in preventing cross-contamination and ensuring the integrity of the cleanroom's environment, playing a key role in the overall contamination control strategy.
We employ a sophisticated differential-pressure management system in our cleanroom to ensure that the pressure in the clean room is higher than outside, so that the air blows out of the clean room when the door is opened.
Particle measurements in cleanroom environments
Particle measurement in cleanroom environments is a critical process in assessing and maintaining the cleanliness of a cleanroom and hereby ensuring the quality and safety of pharmaceuticals, biopharmaceuticals, medical devices, and other products manufactured in these controlled settings. The goal of particle measurement is to monitor and control particulate contamination, which can significantly impact product purity and efficacy.
The choice of particle measurement technique depends on the specific requirements of the cleanroom, including the types of particles of concern, the level of sensitivity required, and the phase in which particles need to be measured (air, surface, or liquid). Together, these techniques provide a comprehensive approach to monitoring and controlling particulate contamination, ensuring that cleanroom environments remain within specified cleanliness levels and supporting the overall goal of protecting product quality and safety.
At Alflow, particle measurement – in accordance with both GMP and ISO Class 7 – is conducted by a third-party firm using state-of-the-art procedures that ensure accuracy and reliability.
Conducting smoke studies for air pattern mapping
Smoke studies, also known as airflow visualization studies, are critical in understanding and validating the airflow patterns within cleanroom environments. These studies are essential for ensuring that the design and operation of a cleanroom effectively control the potential for contamination, protecting the integrity of the manufacturing process and the quality of the products produced. Smoke studies serve to visualize air movement, identify turbulent areas that could harbor or distribute contaminants, and confirm that airflows are as intended.
With the help of smoke studies, we determine where we must take the environmental samples and where we must pay extra attention to cleaning.
Do you want to know more?