Adhering to certifications: Upholding industry standards
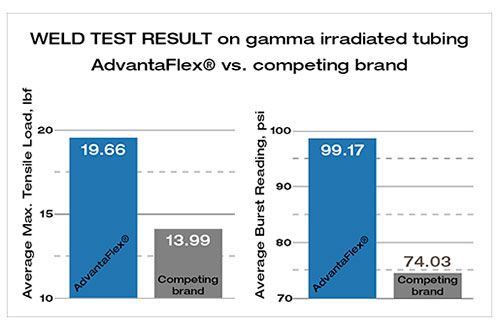
Certification in the cleanroom environment is not just about compliance; it's about upholding a gold standard in operations. Adhering to certifications like ISO 14644 Class 7, GMP (Good Manufacturing Practice) and ISO9001 is a testament to a company's dedication to excellence. These certifications require stringent adherence to quality control, documentation, and process validation, ensuring that we meet industry benchmarks.
The management of a cleanroom facility involves a complex navigation of regulatory requirements. These regulations, often stringent and evolving, dictate everything from construction materials to staff training. For us, staying abreast of these regulations and integrating them into daily operations is crucial. It ensures not just compliance but a commitment to operational excellence and patient safety in the life-science industry.
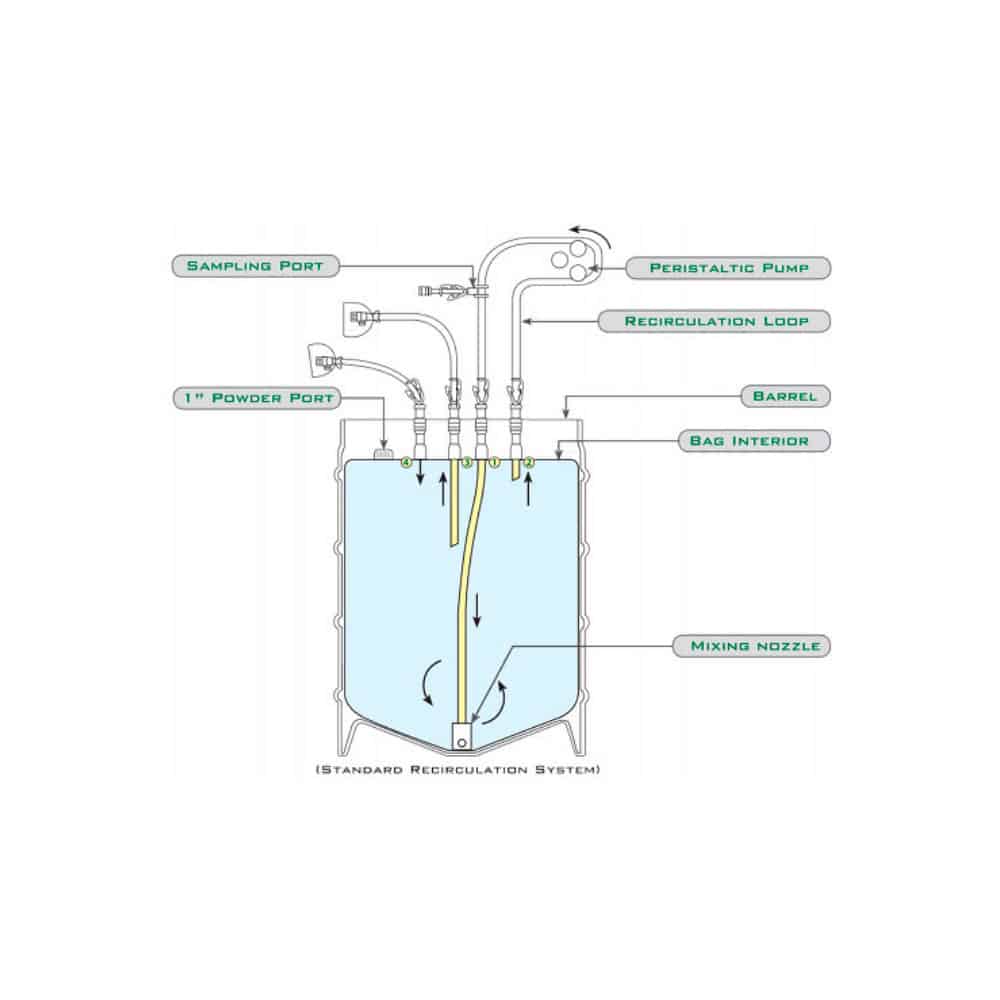
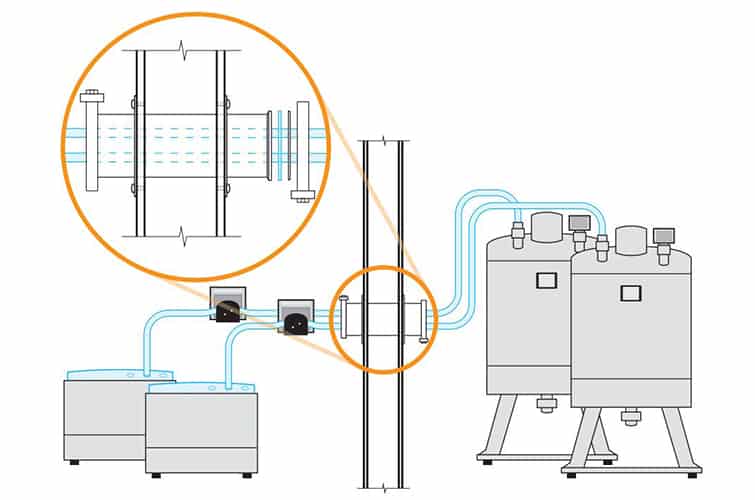
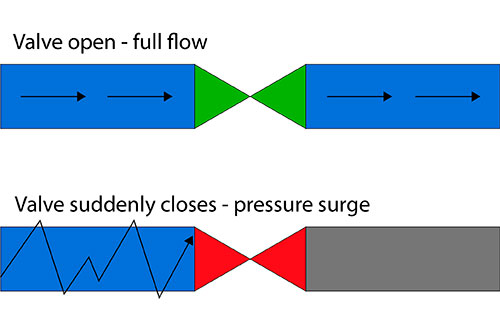
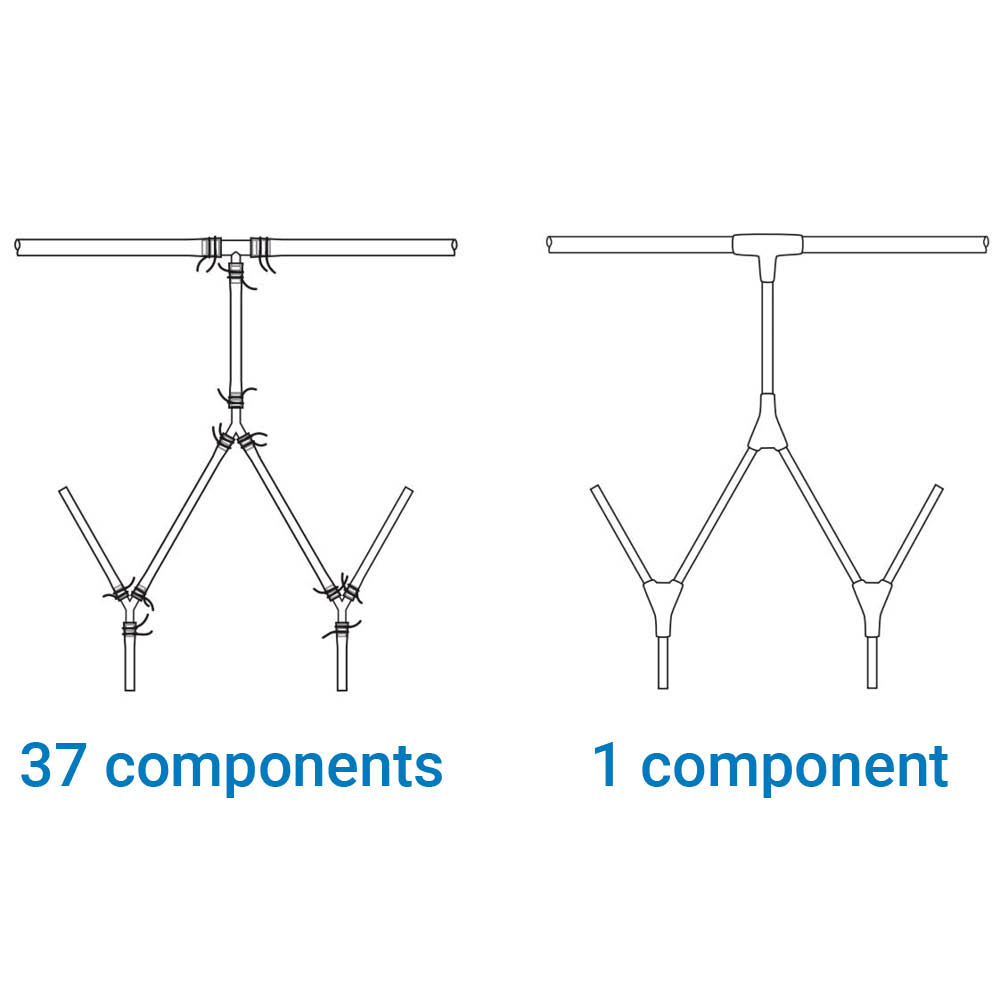
The design of a cleanroom plays a pivotal role in its overall function and efficiency. Factors like air flow, room layout, and material selection directly impact the quality assurance process. A well-designed cleanroom allows for optimal operation while minimizing contamination risks. For us, ensuring that the design aligns with both operational needs and regulatory requirements is key to maintaining the highest standards of quality.
The cleanroom environment is one of continuous improvement, embracing technological advances and process innovations. For us, this means regularly reviewing and updating procedures, investing in new technologies, and training staff in the latest cleanroom practices.
The future of cleanroom technology in the life-science industry is bright, with ongoing advancements in technology and processes. We are at the forefront of this evolution, continuously adapting and improving our cleanroom operations. The commitment to quality, safety, and innovation ensures that the life-science industry will continue to thrive, with cleanroom technology playing a pivotal role in its growth and success.